A piston inside a cylinder undergoes an isentropic compression with the following values.
$latex p_1= 4.57 \frac{N}{cm^2}$
$latex p_2= 34.07 \frac{N}{cm^2}$
$latex V_1 = 257 cm^3$
If the specific heat ratio (k) is 1.4, What is the compressed volume?
(A) 61.2cm3
(B) 30.5cm3
(C) 90.4cm3
(D) 257cm3
A plane intercepts the coordinate axis at (4,6,10). What are the Miller indices for this plane?
(A) (2,3,5) (B) (15,10,6) (C) (-4,-6,-10) (D) (10,6,4)
Correct Answer: B
Which of the following can not be equal to 1 mol of the substance at standard conditions?
(A)20 grams of water (B) 6.02 x 1023 molecules of Uranium (C) 28 grams of N2 (D) 22.4 liters on Argon
Correct Answer: A
More PE Exam Questions
- 1
If a process is considered to be internally reversible and adiabatic, what can the change in entropy be assumed to be?
- 2A plane intercepts the coordinate axis at (4,6,10). What are the Miller indices for this plane?
- 3
What is the oxidation state of Carbon in Carbonate (CO3-2)
- 4How much heat is dissipated when a current of 15 amps passes through a 4 ohm resistor?
- 5
You must choose between four pieces of comparable equipment based on the costs and salvage values given below. All four pieces have a life of 8 years.
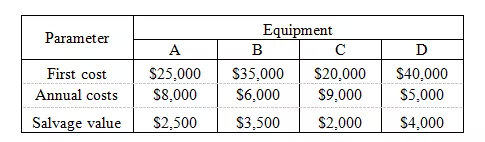
The discount rate is 12%. Ignore taxes. The two most preferable projects and the approximate difference between their present worth values based on least cost are:
